Between Patents and Patients: Humira Case Study
Humira's wide range of indications has earned it the nickname "Swiss army knife" of pharmaceuticals. The success of Humira can be attributed not only to its therapeutic benefits but also to its patent and pricing practices. Over the past two decades, AbbVie has increased the drug's price 30 times.

AbbVie's immunosuppressive drug Adalimumab, commonly known as Humira, has gained fame as a versatile treatment for various debilitating diseases like rheumatoid arthritis, plaque psoriasis, Crohn's disease, and ulcerative colitis. Its wide range of indications has earned it the nickname "Swiss army knife" of pharmaceuticals.
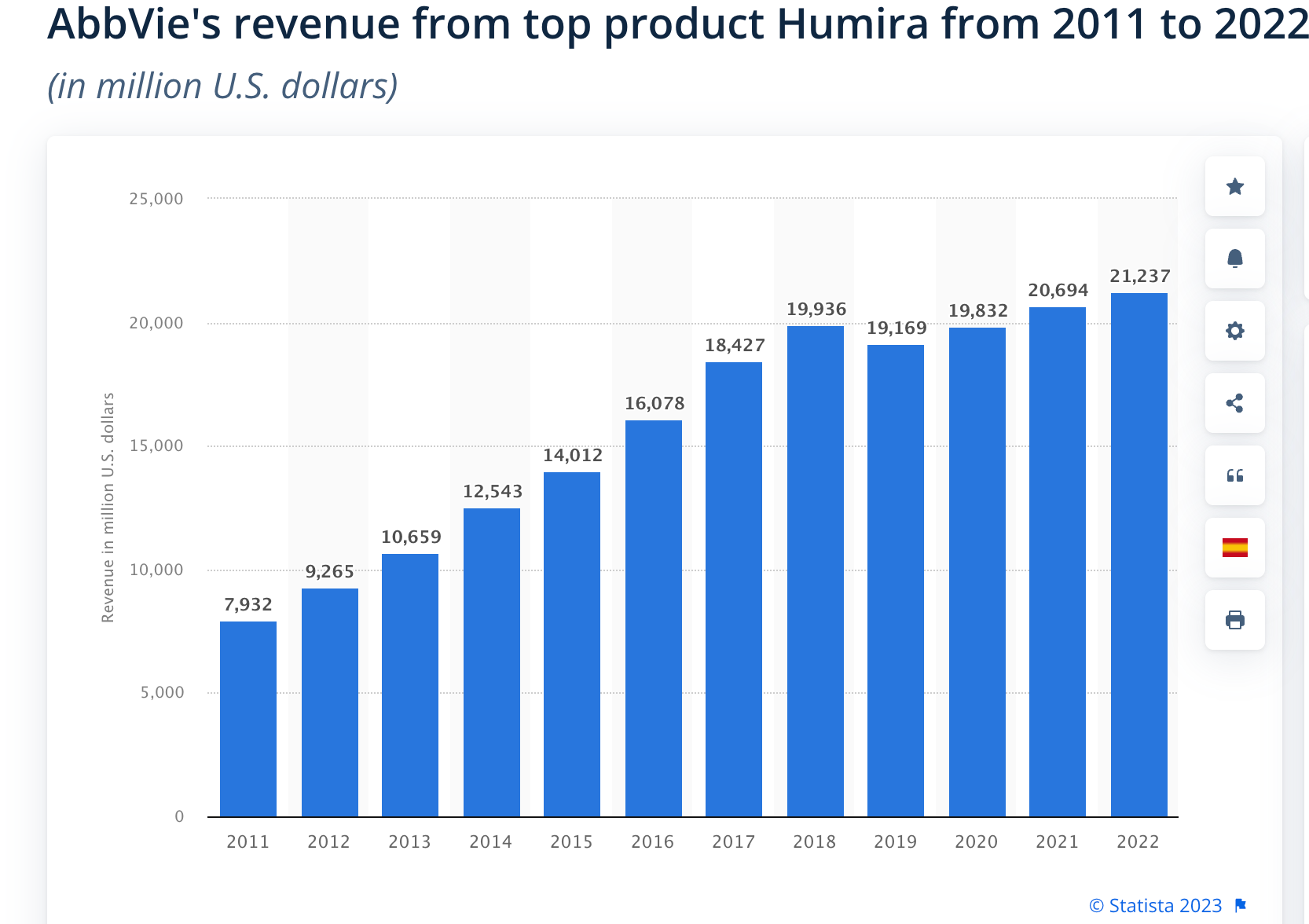
By 2006, the sales of Humira had reached $2 billion, and within a span of two years, it more than doubled. By 2013, Humira had become the top-selling drug globally, generating $10.7 billion in sales across over 60 markets. Remarkably, even this enormous sales figure nearly doubled again in the following five years. In the United States, the average price of Humira doubled from approximately $19,000 per year per patient in 2012 to over $38,000 in early 2018. With millions of patients worldwide, Humira is projected to generate a staggering revenue of $240 billion by 2024. In 2021 alone, Humira generated $20.7 billion in revenue for its manufacturer.
However, the success of Humira can be attributed not only to its therapeutic benefits but also to AbbVie's strategic patent and pricing practices. Over the past two decades, AbbVie has increased the drug's price 30 times, most recently by 8%. The company has meticulously built a fortress of patents around Humira, from filing around 247 patents to securing over 130 patents, it covered every aspect of the drug's manufacturing, administration, formulation, and delivery devices. This extensive patent portfolio made it challenging for competitors to replicate the drug without infringing on AbbVie's intellectual property. Several of these patents were granted in recent years as the expiration date of Humira's primary patent approached in 2016.
Over the years, the U.S. Patent and Trademark Office (USPTO) has granted the company over 30 patents for the administrative methods of Humira, more than 25 patents for different drug formulations, over 50 patents related to manufacturing processes, and approximately 20 patents for delivery devices used by customers. These patents provided an extended protection span for Humira, with the latest expiration date set for 2034.
AbbVie executives defend the high costs of Humira, claiming that they are necessary to support research and development. However, per Committee on Oversight and Reform, 2021 shows that a significant portion of AbbVie's so-called research expenditures has been spent towards safeguarding Humira's market dominance by making "enhancements" to the drug in order to acquire additional patents. The CEO of AbbVie, Richard Gonzalez, has outlined several improvements that the company plans to implement. These enhancements include the development of higher-concentration formulations, the use of smaller needles, the introduction of new dispensing pens, and the implementation of a monthly dosing regimen. But, findings from Phase III clinical trials indicate that these formulation changes primarily led to a reduction in injection site pain, with no statistically significant differences in patient outcomes resulting from the dosing changes. The primary goal of these "enhancements" was to only create barriers for their competitors through patent evergreening and ticketing strategies from letting them enter the market. AbbVie has effectively implemented a highly profitable strategy to maintain its position as the market leader and hinder competition from biosimilars for Humira. By constructing an extensive patent portfolio, focusing on minimal changes to the existing product in their research and development endeavours, and consistently raising prices. Moreover, AbbVie has achieved success in legal battles against biosimilars, effectively delaying their entry into the market until 2023.
Additionally, the report highlights that AbbVie's research expenditures accounted for only a small fraction of its overall revenue. Between 2009 and 2018, AbbVie reportedly spent $5.19 billion on research and development, equivalent to just 7.4% of Humira's net revenue in the United States during the same period.
The validity of secondary patents requires careful consideration of both biological and social factors. While these patents differ from the original in terms of formulation and composition, it becomes equally important to determine their social impacts. Assuming that the patent terms outlined in the Hatch-Waxman Act are optimal and a 20-year exclusivity period is appropriate, the decision to grant extensions becomes nuanced.
From a social cost perspective, maintaining a monopoly on an expensive pharmaceutical like Humira can have negative implications for society's financial well-being. The U.S. insurance system relies on risk pooling, where healthier individuals bear the cost for less healthy individuals through insurance premiums. Continuing a monopoly when lower-cost biosimilars are available and provide similar benefits can increase this financial burden.
Granting extensions would require substantial and novel patient benefits beyond what was observed in clinical trials comparing different formulations of Humira. Addressing the social cost of maintaining an expensive monopoly would require substantial and quantifiable patient benefits. It remains uncertain whether a reduction in injection site pain alone would meet this requirement. AbbVie's strategy with Humira may not be easily replicable by other products. Humira had a favourable market position upon its launch, faced limited competition in the rheumatoid arthritis market, had a unique injectable formulation, and demonstrated effectiveness across multiple indications. These trials showed little improvement in managing rheumatoid arthritis symptoms and disease progression.
AbbVie has aggressively pursued legal action against competitors attempting to introduce biosimilar versions of Humira. In 2016, AbbVie sued Amgen, claiming patent violations and delaying the launch of Amgen's biosimilar until 2023. Similar settlements were reached with nine other manufacturers over the next five years, all agreeing to delay market entry until 2023.
AbbVie had sued for patent infringement, threatening Amgen with claims of infringement on 10 Humira patents and 51 other patents. In September 2017, a settlement was ultimately reached, which resulted in Amgen being granted patent licenses for the global use and sale of Amjevita. Now, Amgen’s Amjevita is set to be the first biosimilar to hit the market in January 2023. Samsung Bioepis followed suit, reaching a settlement with AbbVie that allows it to launch its biosimilar in the U.S. on June 30, 2023. Boehringer Ingelheim initially refused to settle, arguing that AbbVie had intentionally added overlapping and unnecessary patents. However, they eventually agreed to a settlement in May 2019, keeping their product off the market until 2023.
AbbVie's strategy of utilising a complex web of patents, making minimal changes to the product, and continuously increasing prices has effectively defended Humira against biosimilar competition. This approach has allowed Humira to dominate the market for over 20 years and become the world's top-selling drug.
In total, eight FDA-approved Adalimumab biosimilars are now cleared for release in the United States in 2023, with some of these drugs having waited nearly a decade for market entry. The actions of AbbVie and the behaviour surrounding patent strategies and patent ever-greening may face increased scrutiny from regulatory bodies leading to stricter regulations to impose stricter standards for ensuring genuine innovation and fair competition in the pharmaceutical industry.
In India, the issue of patent ever greening has received significant attention, particularly in the pharmaceutical sector. India's patent law includes provisions to prevent the grant of patents for inventions that are mere incremental changes or variations of existing patented products or processes. These provisions are aimed at striking a balance between protecting innovation and ensuring affordable access to essential medicines.
One key provision in Indian patent law that addresses ever greening is Section 3(d) of the Patents Act, 1970. According to this provision, a new form of a known substance, a mere discovery of a new form of a known substance, or a new use for a known substance is not considered patentable unless it exhibits significantly enhanced efficacy over the existing substance. This provision aims to prevent the granting of patents for minor modifications or variations of known substances. The Indian Patent Office and the courts have been vigilant in scrutinising patent applications to prevent ever greening. They assess whether the proposed invention meets the criteria of novelty, inventive step, and industrial applicability and whether it satisfies the requirements of Section 3(d) regarding enhanced efficacy.Several high-profile cases involving pharmaceutical patents have taken place in India, including the famous case of Novartis AG vs Union of India and Ors, where the courts have rejected patent applications or invalidated granted patents based on allegations of ever greening. India's approach to patent ever greening reflects its commitment to balancing the rights of patent holders with the need to ensure affordable access to medicines for its population. By preventing the granting of patents for incremental changes, India aims to encourage genuine innovation and foster competition in the pharmaceutical sector.





